Do your industrial devices fail unexpectedly because of poor power sources? Choosing the wrong battery chemistry causes costly downtime and maintenance headaches for your procurement team.
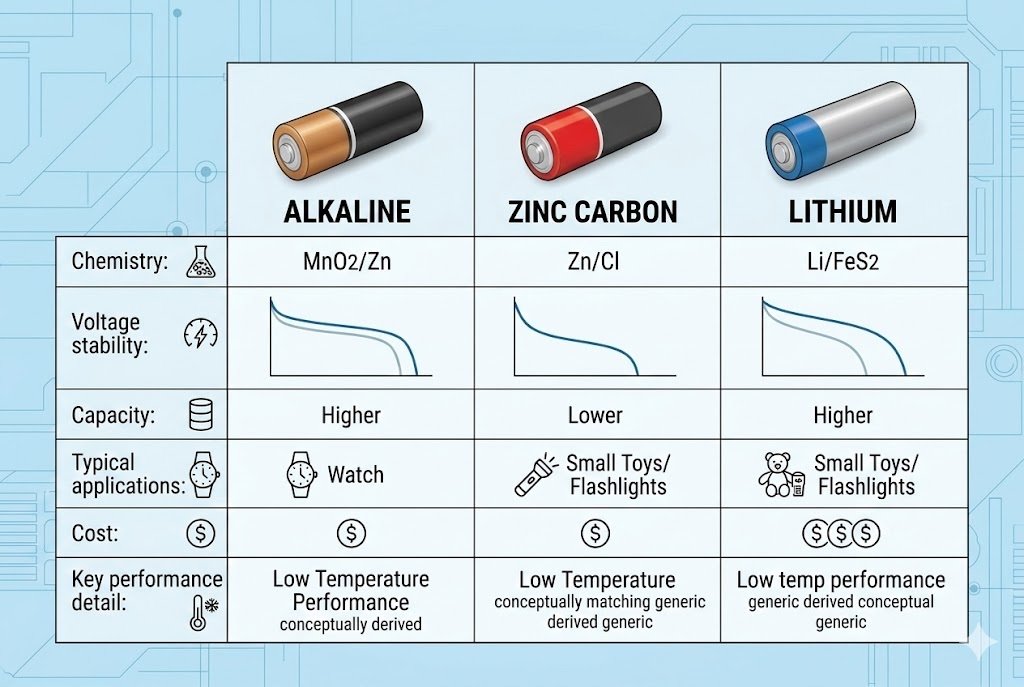
For most high-drain industrial applications, Lithium Iron Disulfide (Li-FeS2)1 offers the best performance with a 20-year shelf life and leakage resistance. However, Alkaline batteries2 remain the cost-effective standard for moderate loads, while Zinc-Carbon is suitable only for low-drain, cost-sensitive clocks and remotes.
I have seen many engineers struggle with this specific choice during my time at Nexcir. We will look at the specific differences between these three common types. Read on to find the right match for your supply chain needs.
Is the Standard Alkaline Battery Still Reliable for Modern Industry?
You likely buy thousands of these, but are they failing your high-power devices? Reliance on old standards can hurt your product's reliability and frustrate your end users.
Alkaline batteries2 rely on a reaction between zinc and manganese dioxide. They offer a balanced energy density and cost ratio, making them the standard for general-purpose electronics, though they struggle with heavy loads and have moderate leakage risks over time.
The Workhorse of the Industry
I deal with many OEM clients who default to Alkaline batteries2 because they are everywhere. They are the middle ground of the battery world. The chemistry uses zinc and manganese dioxide with an alkaline electrolyte, usually potassium hydroxide. This design gives them a higher energy density than the older zinc-carbon types. They work well for devices that need a steady, moderate current. Think of multimeters, flashlights, or electronic door locks.
Voltage Drop Issues3
However, you need to understand their limitations. As an Alkaline battery discharges, its voltage drops gradually. It starts at 1.5V but slides down to 1.0V or lower as it dies. This sloping discharge curve is a problem for modern digital electronics. If your device needs a strict 1.3V to run the processor, the battery might still have 30% capacity left, but the voltage is too low to use it. This results in waste.
The Leakage Problem4
The biggest issue I see is leakage. The electrolyte is corrosive. When the battery discharges, it produces hydrogen gas. If the pressure gets too high, the seal breaks, and the chemical leaks out. This ruins the battery contacts and can destroy your expensive equipment. I always tell my clients: if the device sits in storage for a long time, Alkaline is a risk.
| Feature | Performance Level |
|---|---|
| Nominal Voltage5 | 1.5V |
| Capacity6 | 1800-2800 mAh |
| Shelf Life7 | 5-10 Years |
| High Load Performance | Moderate |
| Leakage Risk8 | Moderate to High |
Should You Ever Choose Zinc-Carbon Batteries for Industrial Equipment?
Price is important, but cheap components can ruin expensive hardware. Using heavy-duty zinc batteries might save pennies but cost dollars in repairs and brand damage.
Zinc-Carbon batteries9, often labeled "Heavy Duty," use an ammonium chloride or zinc chloride electrolyte. They have low energy density and are prone to leakage, making them suitable only for very low-drain devices like wall clocks or simple remote controls.
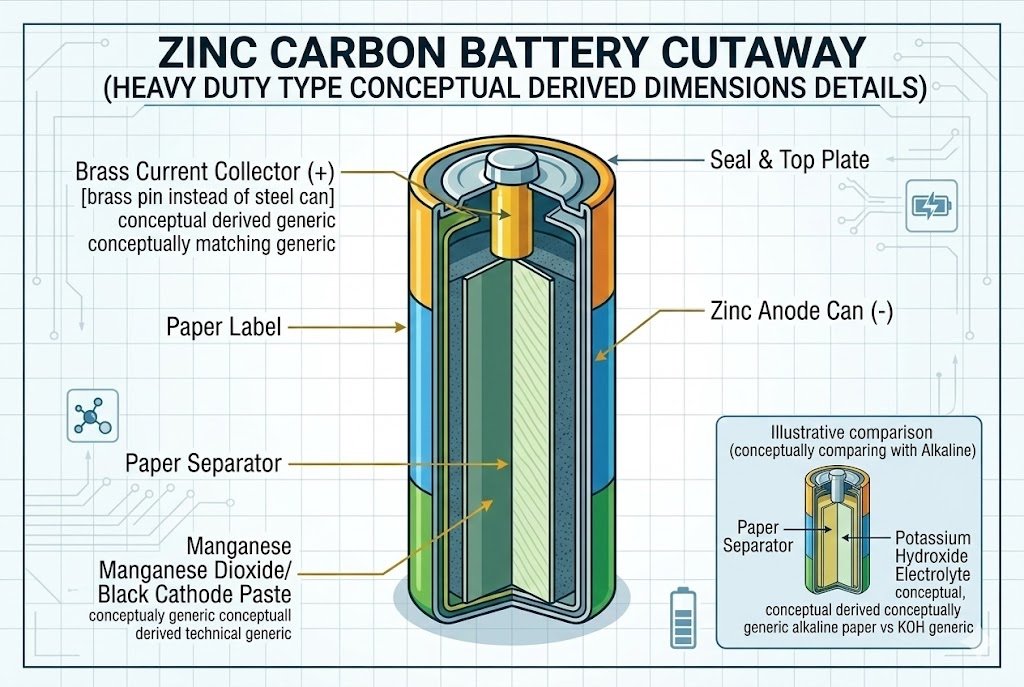
Understanding the "Heavy Duty" Label
Do not let the name fool you. Manufacturers often label Zinc-Carbon batteries9 as "Heavy Duty" or "Super Heavy Duty." In my experience, this is just marketing. Compared to modern standards, they are light duty. This is the oldest dry cell technology. The can itself is made of zinc and acts as the anode. As the battery works, the acid attacks the zinc can.
Why They Fail
The walls of the battery actually get thinner as you use it. This is a fundamental flaw. Once the wall becomes too thin, holes appear. The acidic electrolyte leaks out and eats the metal springs in your battery compartment. I have seen this happen countless times in older equipment. The capacity is also very low. They hold less than half the energy of an Alkaline battery.
When to Use Them
So, why do we still sell them at Nexcir? Cost. They are incredibly cheap. If you are manufacturing a simple device that draws almost no power, like a TV remote or a wall clock, they are fine. For these low-drain applications, the battery will last a year or two. But for anything with a motor, a screen, or a light, you must avoid them. They cannot supply the current fast enough, and the voltage collapses immediately.
| Feature | Zinc-Carbon | Zinc-Chloride (Heavy Duty) |
|---|---|---|
| Electrolyte | Ammonium Chloride | Zinc Chloride |
| Capacity6 | 400-900 mAh | 1000-1500 mAh |
| Leakage Risk8 | Very High | High |
| Best Use Case | Wall Clocks | Remotes |
Is Lithium Iron Disulfide the Ultimate Solution for High-Performance Needs?
Frequent battery replacements drive up labor costs significantly. Ignoring advanced lithium chemistry means missing out on extended lifecycles and superior temperature handling.
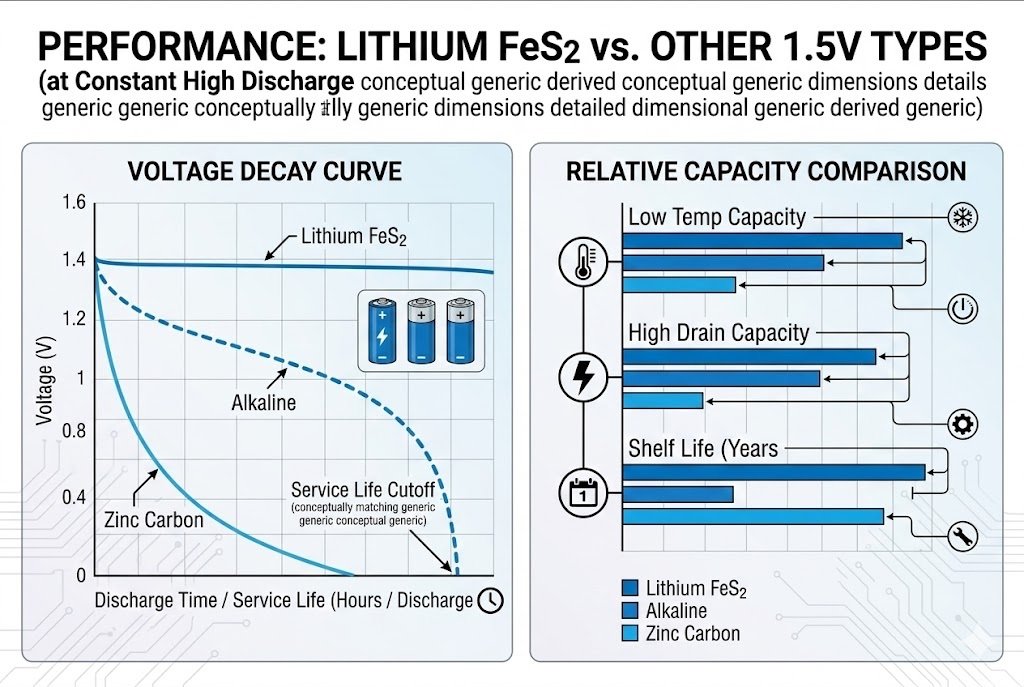
1.5V Lithium batteries (Li-FeS2) provide superior energy density and operate effectively in extreme temperatures ranging from -40°C to 60°C. They are lightweight, leak-proof, and ideal for high-drain devices like cameras, sensors, and medical equipment.
Superior Chemistry10
This is the technology I recommend for critical applications. Please note, I am talking about primary (non-rechargeable) Lithium Iron Disulfide, not the 3.7V Lithium-Ion batteries used in phones. These are direct replacements for standard AA and AAA batteries. The chemistry is totally different from Alkaline. They use lithium metal for the anode and iron disulfide for the cathode.
The Voltage Advantage11
The most impressive feature is the discharge curve. Unlike Alkaline batteries2 that slowly lose voltage, Lithium batteries stay near 1.5V for almost their whole life. They drop off sharply only at the very end. This means your high-tech sensors or cameras run at full power until the last minute. You utilize nearly 100% of the stored energy.
Built for Extremes12
I also recommend these for outdoor equipment. Alkaline batteries2 freeze and stop working in winter. Lithium batteries work fine at -40°C. They are also much lighter, weighing about 30% less than Alkaline cells. This matters for portable handheld devices. The construction involves a spiral wound design inside, which provides a large surface area. This allows them to release energy very quickly without overheating.
The Leak-Proof Design13
Safety is the final selling point. These batteries rarely leak. They do not generate gas in the same way Alkaline cells do. For medical devices or expensive field gear where failure is not an option, the higher price of Lithium is worth it. It is an insurance policy for your hardware.
| Feature | Performance Level |
|---|---|
| Nominal Voltage5 | 1.5V (1.7V Open Circuit) |
| Capacity6 | 3000 mAh+ |
| Shelf Life7 | 15-20 Years |
| Temperature Range14 | -40°C to +60°C |
| Weight | ~15g (AA size) |
How Do Discharge Rates15 and Leakage Risk8s Impact Your Procurement Strategy?
A leaking battery destroys the device and your brand reputation. Failing to account for long-term storage risks is a supply chain disaster waiting to happen.
Discharge rates determine how fast a battery releases energy without voltage drop, while leakage risks depend on the electrolyte stability. Lithium offers the safest long-term storage, whereas Alkaline requires careful rotation to prevent corrosive damage to terminals.

Analyzing Discharge Rates15
When we talk about discharge rate, we are talking about internal resistance. Think of it like a water pipe. Zinc-Carbon has a narrow pipe; water (energy) flows slowly. If you try to force it, the pressure drops. Lithium has a very wide pipe. You can draw a lot of current very fast, and the voltage stays strong. For my B2B clients, I suggest looking at the device's "pulse" requirement. Does your device wake up, send a strong signal, and sleep? That is a high pulse. Alkaline batteries2 recover slowly from high pulses. Their voltage dips, and the device might think the battery is dead. Lithium handles these pulses effortlessly.
The Mechanism of Leakage16
We need to look deeper at why leakage happens to protect your assets. In Alkaline batteries2, leakage is usually potassium hydroxide. This is a strong base that causes chemical burns and eats copper contacts. It happens because the battery produces hydrogen gas as it discharges. If the seal is poor, or if the battery gets hot, the gas pushes the liquid out. Even worse, if you mix old and new batteries, the new one forces energy into the old one (reverse charging). This guarantees a leak. Lithium cells use a non-aqueous electrolyte and have a safety vent. They are chemically much more stable.
Strategic Recommendations for Buyers17
Based on my 20 years in this industry, here is my advice for your procurement strategy:
- For Low Value, Low Drain: If you make remote controls, stick to Zinc-Carbon or low-end Alkaline to keep the Bill of Materials (BOM) low.
- For General Consumer Goods: Use decent Alkaline brands. But, advise the user to remove them if not used for months.
- For Industrial/Medical/Outdoor: Do not compromise. Use Lithium (Li-FeS2). The cost of one service call to fix a corroded terminal costs more than 100 batteries.
- Storage: If you stock spare parts, remember shelf life. Zinc-Carbon dies in 2 years. Alkaline lasts 5-7 years. Lithium stays fresh for 20 years. This affects your inventory waste.
Summary of Risks
| Chemistry | High Drain Risk18 | Leakage Risk8 | Storage Risk19 |
|---|---|---|---|
| Zinc-Carbon | Device will shut down | High (Acidic) | High (Short Shelf Life7) |
| Alkaline | Voltage sag | Moderate (Caustic) | Moderate |
| Lithium | Low | Very Low | Very Low |
Conclusion
Choosing the right 1.5V battery balances cost, performance, and risk; Lithium is best for industry, Alkaline for general use, and Zinc-Carbon only for the lowest power needs.
Explore how Li-FeS2 batteries offer superior performance, long shelf life, and leakage resistance for high-drain industrial applications. ↩
Learn about the limitations and risks of using Alkaline batteries in high-power devices, including voltage drop and leakage issues. ↩
Discover why voltage drop in Alkaline batteries can lead to wasted energy and device malfunction in modern electronics. ↩
Find out how leakage from Alkaline batteries can damage equipment and learn strategies to prevent it. ↩
Learn about the nominal voltage of batteries and how it affects device compatibility and performance. ↩
Discover how battery capacity impacts energy storage and device operation, guiding your selection process. ↩
Understand the importance of battery shelf life in procurement strategies and long-term storage solutions. ↩
Compare leakage risks across battery types to make informed decisions for device safety and longevity. ↩
Understand the cost benefits and limitations of Zinc-Carbon batteries for low-drain devices like clocks and remotes. ↩
Learn about the advanced chemistry of Li-FeS2 batteries and why they are recommended for high-performance needs. ↩
Understand how Lithium batteries provide consistent voltage, ensuring full power for high-tech devices until depletion. ↩
Explore the benefits of Lithium batteries in extreme temperatures and their lightweight design for portable devices. ↩
Learn about the safety features of Lithium batteries that minimize leakage risks, making them ideal for sensitive equipment. ↩
Explore the temperature resilience of Lithium batteries, making them suitable for outdoor and industrial use. ↩
Discover how discharge rates affect energy release and device functionality, especially in high-drain applications. ↩
Understand the chemical processes behind battery leakage and explore methods to protect your devices. ↩
Get expert advice on selecting the right battery type for various applications to optimize performance and cost. ↩
Explore why Zinc-Carbon batteries are unsuitable for high-drain applications and the potential device shutdown risks. ↩
Learn about the shelf life of different battery types and how it impacts inventory waste and management. ↩